Lean Validation
FOR THE LIFE SCIENCE INDUSTY
To faciliate lean validation best practices, we leverage our flagship application ValidationMaster™ – the most ADVANCED Enterprise Validation Management and Quality system on the market. We leverage ValidationMaster through each service enagement to streamline test script development and execution, requirements management, incident management, electronic review and approval, risk management, cybersecurity qualification (CyQ) and the delivery of validation reports in your unique format.
LEAN VALIDATION STRATEGIES
For 503A/503B Facilities
The Compound Pharmacy market sector is in the midst of adapting to recent regulatory changes imposed by the U.S. FDA. Section 503A of the U.S. FD&C Act provides governance for the conditions that must be satisfied for human drug products compounded by a licensed pharmacist in a State-licensed pharmacy or Federal facility or by a licensed physician, to be exempt from three key sections of the FD&C Act. Section 503B is relatively new and creates a new entry point for medications into the drug supply chain through outsourcing facilities.
Outsourcing facilities can be used to fill the gap between traditional pharmacy compounding and industrial manufacturing where compounded products are needed for administration by health care facilities.
One of the most significant changes for outsourcing facilities is that they now must comply with cGMPs applicable to drug manufacturers. Although 503A facilities are exempt from cGMP, they are still subject to USP compliance. From a validation perspective, any computer systems deployed by 503B companies are subject to FDA computer systems guidelines.
We’ve worked with 503B Facilities
Helping them understand validation requirements and apply lean validation techniques to enterprise computer systems that drive cGMP and quality processes.
Mastering Regulatory Compliance
and Sustained Quality
Mastering Regulatory Compliance means going beyond the basic rudiments of in the execution of your processes to a higher level of mastery and best practices. Pharmaceutical and healthcare companies are adopting advanced cloud-based technologies, the Internet of Things (Iot), mobility and many other technologies at an increasing pace. The challenges associated with cGxP compliance and other predicate rule requirements are now coupled with issues concerning cybersecurity and data integrity.
The principles of Validation and verification for computer systems endures and yet many pharmaceutical and healthcare companies are still conducting manual validation exercises on paper and using non-automated workflow processes to review and approve validation deliverables.
Lean validation for pharmaceutical & healthcare companies
We practice LEAN VALIDATION. OnShore Technology Group, Inc. understands the unique requirements facing today’s pharmaceutical companies and we are ever-seeking new ways to address the unique challenges of this market sector. Lean validation strategies are designed to automate the validation process while eliminating wasteful, manual processes.
Going Lean in Pharma and Healthcare
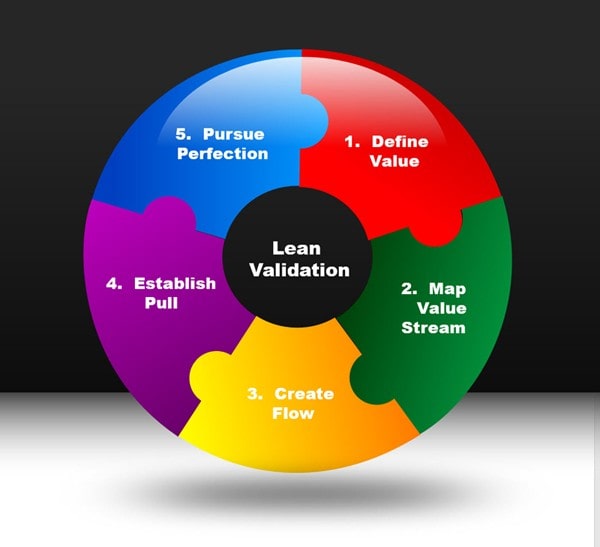
Lean validation is derived from the lean manufacturing process. Simply put, lean manufacturing is the process of eliminating waste, minimizing risk and driving quality, efficiency and compliance. A common misconception is that lean is suited only for manufacturing. This is not true. Lean principles may be applied to any process that would benefit from the elimination of wasteful activities and process improvement. Lean validation thus is not a tactic or a cost reduction program, but a way of thinking and acting for today’s validation team. The figure below depicts the overall strategy for lean validation.
There are five principles of lean validation derived from lean manufacturing principles. Lean validation is powered through people, processes and technology. Automation drives lean validation processes.
Principle 1 – VALUE – Lean thinking in manufacturing begins with a detailed understanding of what value the customer assigns to product and services. Lean thinking from an independent validation and verification perspective begins with a detailed understanding of the goals and objectives of the validation process and its adherence to compliance objectives. The principle of VALUE requires the validation team to therefore focus on the elimination of waste to deliver the value the end customer (your organization) in the most cost-effective manner. The computer systems validation process is designed for the purpose of assuring that software applications meet their intended use. The value derived from the validation process is greater software quality, enhanced ability to identify software defects as a result of greater focus and elimination of inefficient and wasteful processes. AUTOMATION IS THE FOUNDATION THAT FACILITATES THE ACHIEVEMENT OF THIS VALUE PRINCIPLE.
Principle 2 – VALUE STREAM – The value stream, from a lean perspective is the comprehensive product life-cycle from the raw materials through customer’s end use, and ultimate disposal the product. To effectively eliminate waste, the ultimate goal of lean validation, there must be an accurate and complete understanding of the value stream. Validation processes must be examined end-to-end to determine what value is added to the objective of establishing software quality and compliance. Any process that does not add value to the validation process should be eliminated. We recommend value stream mapping for the validation process to understand where value is added and where non-value -added processes can be eliminated. Typical “Muda” or wastes commonly revealed from validation process mapping are:
- Wasteful Legacy Processes (“we have always done it this way”)
- Processes That Provide No Value To Software Quality At All
- Manual Process Bottlenecks That Stifle Processes
Principle 3 – FLOW – The lean manufacturing principle of flow is about creating a value chain with no interruption in the production process and a state where each activity is fully in step with every other. A comprehensive assessment and understanding of flow throughout the validation process is essential to the elimination of waste. From a validation perspective, optimal flow is created through the process when, for example, users have the ability to automatically create requirements from test scripts for automated traceability thereby eliminating the process of manually tracing each test script to a requirement. Another example is when a user has the ability to navigate through a software application and the test script process is automatically generated. Once generated, it is automatically published to a document portal where it is routed electronically for review and approval. All of this requires AUTOMATION to achieve the principle of FLOW. For process optimization and quality control throughout the validation lifecycle, information should optimally flow throughout the validation process in an efficient manner minimizing process and document bottlenecks with traceability throughout the process.
Principle 4 – PULL – A pull system is a lean manufacturing is used to reduce waste in the production process. Components used in the manufacturing process are only replaced once they have been consumed so companies only make enough products to meet customer demand. There is much waste in the validation process. The PULL strategy for validation may be used to reduce wastes such as duplication of effort, streamlining test case development and execution, electronic signature routing/approval and many others. Check out our blog “The Validation Post” for more information.
Principle 5 – PERFECTION – Validation processes are in constant pursuit of continuous improvement. Automation is KEY. Lean validation engineers and quality professional relentlessly drive for perfection. Step-by-step validation engineers must identify root causes of software issues, anomalies, and quality problems that affect the suitability of a system for production use. As computing systems environments evolve and become more complex and integrated, validation engineers must seek new, innovative ways to verify software quality and compliance in today’s advanced systems. Perfection cannot easily be achieved through manual processes.
Accelerating Validation of
Advanced Enterprise Solutions
Technology companies are delivering more innovative solutions to their clients that drive productivity, efficience, governance and compliance to levels never previously achieved.
Technology software in particular players large and small are driven help companies achieve sustained compliance and manage information in a manner that ensures data integrity, authenticity and security over time. As enterprise technologies such as SAP, Microsoft Azure, Dynamics 365, Oracle e-business, BatchMaster, Merit Solutions MAXLife and even players such as Intel are deployed within life sciences systems environments, these systems require independent validation and verification (IV&V) to provide software assurance and quality. Outsourcing the IV&V process is the very latest trend among life sciences companies. The efficiencies and independence gained through outsourcing have led many companies to execute this important business strategy.
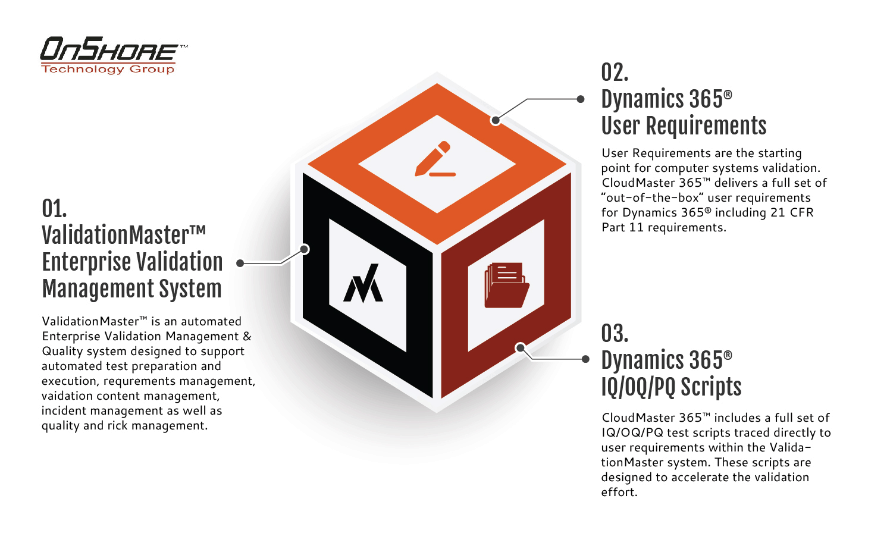
Validation accelerators help deliver value, efficiency, and compliance
Although validation mandates have been with us for years, many technology companies struggle with how to present their solutions to quality and technology life sciences officials as “validation friendly”. The introduction of cloud solutions, mobile technology integrations, the Internet of Things (IoT) and other technologies have caused software companies to rethink their validation strategies. An example below is for Microsoft Dynamics 365. We offer more accelerators for other advanced technology solutions.
Powering Speciality Pharma
Automation and Compliance
The Speciality Pharma industry sector is growing globally at a rapid pace. The promise of their new therapies offer tremendous promise for the treatment of chronic conditions in large populations. Innovation is key to the ultimate sustained success of this market sector.
Quality and compliance are at the core of everything in the specialty pharma industry. Patient engagement also increases their changes of regulatory and commerical success. Enterprise technologies such as ERP systems, quality management systems, and learning management systems play an important role in driving quality and sustained compliance.
OnShore Technology Group, Inc. offers unique solutions that help specialty pharma companies implement lean validation processes. We offer automated solutions for Enterprise Validation Management and Quality, learning and competency management solutions and professional services to drive your success.
Powering Lean Validation in Specialty Pharma
Lean validation is derived from the lean manufacturing process. Simply put, lean manufacturing is the process of eliminating waste, minimizing risk and driving quality, efficiency and compliance. A common misconception is that lean is suited only for manufacturing. This is not true. Lean principles may be applied to any process that would benefit from the elimination of wasteful activities and process improvement. Lean validation thus is not a tactic or a cost reduction program, but a way of thinking and acting for today’s validation team. The figure below depicts the overall strategy for lean validation.
As highlighted above, lean validation requires automation. ValidationMaster™ powers lean validation and is designed to facilitate automation throughout the process. The system manages the full lifecycle of validation processes and is designed to eliminate traditional manual, paper-based processes.
Advancing Quality and Lean Validation
in the Chemical Industry
As one of the largest manufacturing industries in the United States, the chemical market sector serves an expansive domestic and global market. Chemical companies have a great responsibility to the public and regulators to ensure that their chemicals are responsiblity managed while under their charge. Chemical companies have complex manufacturing, marketing and sales, R&D and other key data-driven processes that provide opportunities for productivity enhancements and process improvment.
Like many other industries, chemical companies must do optimize their production footprints and redesign processes to ensure sustained compliance. Companies are seeking ways toi better manage and track inventory, improve supply chain processes and validate computer systems to ensure compliance and quality.
OnShore delivers advance independent validation and verification (IV&V) services for the chemical industry. Our solutions for the chemical industry include Enterprise Validation Management, competency and learning management and quality.
Lean Validation Best Practices
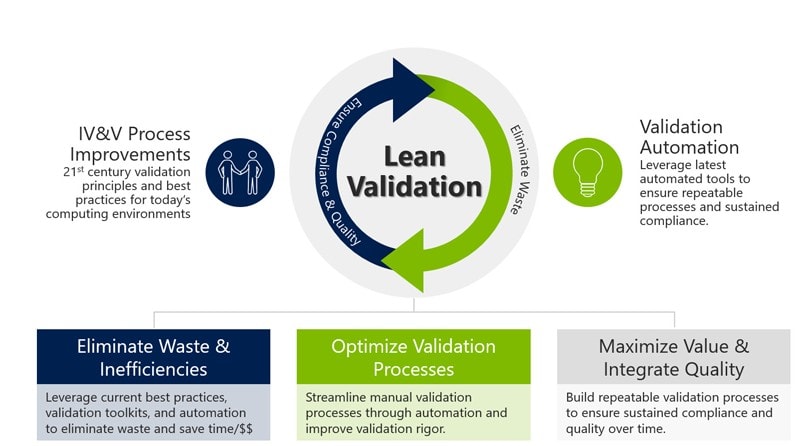
OnShore Technology Group, Inc. is a pioneer in lean validation and has developed principles and best practices to support lean validation processes. To power our lean processes, we leverage ValidationMaster™, an Enterprise Validation Management system exclusively designed to facilitate lean validation and automate the validation process.
Lean validation CANNOT be achieved without automation. Lean validation processes leverage advanced technology designed to fulfill the technical, business and compliance requirements for software validation eliminating the use of manual, paper-based processes. Optimized validation processes are deployed using the latest global best practices to ensure the right amount of validation rigor based on critical quality attributes, risks, Cybersecurity and other factors.
Lean Validation & Verification (IV&V)
For Biotechs
In the biotech market sector, growth depends on new discoveries for the treatment of diseases. On the whole, the biotechnology industry is one of the most research-intensive sectors in the world. As they advance therapies designed to help patients, they must comply with quality and compliance mandates.
Computer systems used by biotechnology firms must be validated in accordance with global regulatory guidelines. Many companies are pre-market and may not have the requisite procedures in place to support validation.
OnShore helps biotechnology companies establish policies and procedures to help ensure compliance. We offer lean validation solutions that drive operational efficiences and streamline the validation process.
Lean Thinking in Action For Biotechs
OnShore Technology Group, Inc. is a pioneer in lean validation and has developed principles and best practices to support lean validation processes. To power our lean processes, we leverage ValidationMaster™, an Enterprise Validation Management system exclusively designed to facilitate lean validation and automate the validation process.
Lean validation CANNOT be achieved without automation. Lean validation processes leverage advanced technology designed to fulfill the technical, business and compliance requirements for software validation eliminating the use of manual, paper-based processes. Optimized validation processes are deployed using the latest global best practices to ensure the right amount of validation rigor based on critical quality attributes, risks, Cybersecurity and other factors.
Powering Lean Validation For
Medical Device Companies
Today’s forward-thinking medical device manufacturers must overcome significant challenges to achieve success in an expanding global marketplace. A recent study suggested that the industry is projected to grow at a CAGR of 4.6 percent from 2017 to 2021. While the industry trends are positive, device manufacturers must be ready to deliver and bring their devices to a competitive market in the most cost-effective manner.
To achieve their goals, medical device manufacturers are adopting advanced, cloud-based technologies at a rapid pace for quality management, supply chain, content management and many other regulated business processes. Device manufacturers understand all too well that they must ultimately reduce time to market while shortening product life cycles. Quality and innovation are critical to their ultimate success. When validating computer systems during the implementation and deployment of state-of-the-art technology, time is of the essence. Medical device companies are beginning to THINK LEAN to address their challenges. Traditional, manual, paper-based processes can be arduous and costly for most manufacture’s given the speed at which technology is advancing.
Validation as we know it is DEAD. The most innovative and progressive medical device companies are GOING LEAN. Given the challenges of cybersecurity, data integrity, and electronic records management, companies are shifting to a more lean, agile validation process which will drive greater efficiencies and sustained compliance.
Lean Thinking For Medical Device
Lean validation is derived from the lean manufacturing process. Simply put, lean manufacturing is the process of eliminating waste, minimizing risk and driving quality, efficiency and compliance. A common misconception is that lean is suited only for manufacturing. This is not true. Lean principles may be applied to any process that would benefit from the elimination of wasteful activities and process improvement. Lean validation thus is not a tactic or a cost reduction program, but a way of thinking and acting for today’s validation team. The figure below depicts the overall strategy for lean validation.
There are five principles of lean validation derived from lean manufacturing principles. Lean validation is powered through people, processes and technology. Automation drives lean validation processes.
Principle 1 – VALUE – Lean thinking in manufacturing begins with a detailed understanding of what value the customer assigns to product and services. Lean thinking from an independent validation and verification perspective begins with a detailed understanding of the goals and objectives of the validation process and its adherence to compliance objectives. The principle of VALUE requires the validation team to therefore focus on the elimination of waste to deliver the value the end customer (your organization) in the most cost-effective manner. The computer systems validation process is designed for the purpose of assuring that software applications meet their intended use. The value derived from the validation process is greater software quality, enhanced ability to identify software defects as a result of greater focus and elimination of inefficient and wasteful processes. AUTOMATION IS THE FOUNDATION THAT FACILITATES THE ACHIEVEMENT OF THIS VALUE PRINCIPLE.
Principle 2 – VALUE STREAM – The value stream, from a lean perspective is the comprehensive product life-cycle from the raw materials through customer’s end use, and ultimate disposal the product. To effectively eliminate waste, the ultimate goal of lean validation, there must be an accurate and complete understanding of the value stream. Validation processes must be examined end-to-end to determine what value is added to the objective of establishing software quality and compliance. Any process that does not add value to the validation process should be eliminated. We recommend value stream mapping for the validation process to understand where value is added and where non-value -added processes can be eliminated. Typical “Muda” or wastes commonly revealed from validation process mapping are:
- Wasteful Legacy Processes (“we have always done it this way”)
- Processes That Provide No Value To Software Quality At All
- Manual Process Bottlenecks That Stifle Processes
Principle 3 – FLOW – The lean manufacturing principle of flow is about creating a value chain with no interruption in the production process and a state where each activity is fully in step with every other. A comprehensive assessment and understanding of flow throughout the validation process is essential to the elimination of waste. From a validation perspective, optimal flow is created through the process when, for example, users have the ability to automatically create requirements from test scripts for automated traceability thereby eliminating the process of manually tracing each test script to a requirement. Another example is when a user has the ability to navigate through a software application and the test script process is automatically generated. Once generated, it is automatically published to a document portal where it is routed electronically for review and approval. All of this requires AUTOMATION to achieve the principle of FLOW. For process optimization and quality control throughout the validation lifecycle, information should optimally flow throughout the validation process in an efficient manner minimizing process and document bottlenecks with traceability throughout the process.
Principle 4 – PULL – A pull system is a lean manufacturing is used to reduce waste in the production process. Components used in the manufacturing process are only replaced once they have been consumed so companies only make enough products to meet customer demand. There is much waste in the validation process. The PULL strategy for validation may be used to reduce wastes such as duplication of effort, streamlining test case development and execution, electronic signature routing/approval and many others. Check out our blog “The Validation Post” for more information.
Principle 5 – PERFECTION – Validation processes are in constant pursuit of continuous improvement. Automation is KEY. Lean validation engineers and quality professional relentlessly drive for perfection. Step by step validation engineers must identify root causes of software issues, anomalies, and quality problems that affect the suitability of a system for production use. As computing systems environments evolve and become more complex and integrated, validation engineers must seek new, innovative ways to verify software quality and compliance in today’s advanced systems. Perfection cannot easily be achieved through manual processes.
Lean Thinking In
Nutraceuticals
Nutraceuticals are products that provide health and medicinal benefits, including the prevention and treatment of diseases in addition to the basic nutritional value found in foodstuff. The nutraceuticals industry is in a period of rapid growth in the U.S. and around the globe. As the globalization of manufacturing and distribution continues, the need for stronger systems to assess and ensure quality in dietary supplements becomes more pressing. Some manufacturers in the nutraceutical industry have had challenges with product and supply chain integrity and are adopting Enterprise Resource Planning (ERP) systems to address these challenges.
Since 2010, nutraceutical companies are subject to cGMP regulatory guidelines, thus any systems deployed that manage GMP processes must be validated. Given the market imperatives for greater efficiency and optimization, nutraceuticals are GOING LEAN.
Lean Validation Best Practices
OnShore Technology Group, Inc. is a pioneer in lean validation and has developed principles and best practices to support lean validation processes. To power our lean processes, we leverage ValidationMaster™, an Enterprise Validation Management system exclusively designed to facilitate lean validation and automate the validation process.
Lean validation CANNOT be achieved without automation. Lean validation processes leverage advanced technology designed to fulfill the technical, business and compliance requirements for software validation eliminating the use of manual, paper-based processes. Optimized validation processes are deployed using the latest global best practices to ensure the right amount of validation rigor based on critical quality attributes, risks, Cybersecurity and other factors.
Mastering Regulatory
Compliance and Quality
The vaccine industry is thriving yet going through multiple challenges in its mission to take a candidate product to the clinic and eventually to market. In the current vaccine-manufacturing environment, time to market and cost effectiveness are key issues that must be sufficiently addressed to deliver shareholder and value to its patients.
To achieve its business objectives, the vaccine industry is adopting advanced enterprise technologies to facilitate core business processes including validation. These state-of-the-art technologies design to simplify vaccine development and manufacturing and streamline the validation process are becoming ever-more crucial. The vaccine industry is going lean. Forward-thinking vaccine manufacturers are leveraging lean validation processes to help ensure quality and reduce time.
Lean Thinking For Vaccine Industry
Lean validation is derived from the lean manufacturing process. Simply put, lean manufacturing is the process of eliminating waste, minimizing risk and driving quality, efficiency and compliance. A common misconception is that lean is suited only for manufacturing. This is not true. Lean principles may be applied to any process that would benefit from the elimination of wasteful activities and process improvement. Lean validation thus is not a tactic or a cost reduction program, but a way of thinking and acting for today’s validation team. The figure below depicts the overall strategy for lean validation.
There are five principles of lean validation derived from lean manufacturing principles. Lean validation is powered through people, processes and technology. Automation drives lean validation processes.
Principle 1 – VALUE – Lean thinking in manufacturing begins with a detailed understanding of what value the customer assigns to product and services. Lean thinking from an independent validation and verification perspective begins with a detailed understanding of the goals and objectives of the validation process and its adherence to compliance objectives. The principle of VALUE requires the validation team to therefore focus on the elimination of waste to deliver the value the end customer (your organization) in the most cost-effective manner. The computer systems validation process is designed for the purpose of assuring that software applications meet their intended use. The value derived from the validation process is greater software quality, enhanced ability to identify software defects as a result of greater focus and elimination of inefficient and wasteful processes. AUTOMATION IS THE FOUNDATION THAT FACILITATES THE ACHIEVEMENT OF THIS VALUE PRINCIPLE.
Principle 2 – VALUE STREAM – The value stream, from a lean perspective is the comprehensive product life-cycle from the raw materials through customer’s end use, and ultimate disposal the product. To effectively eliminate waste, the ultimate goal of lean validation, there must be an accurate and complete understanding of the value stream. Validation processes must be examined end-to-end to determine what value is added to the objective of establishing software quality and compliance. Any process that does not add value to the validation process should be eliminated. We recommend value stream mapping for the validation process to understand where value is added and where non-value -added processes can be eliminated. Typical “Muda” or wastes commonly revealed from validation process mapping are:
- Wasteful Legacy Processes (“we have always done it this way”)
- Processes That Provide No Value To Software Quality At All
- Manual Process Bottlenecks That Stifle Processes
Principle 3 – FLOW – The lean manufacturing principle of flow is about creating a value chain with no interruption in the production process and a state where each activity is fully in step with every other. A comprehensive assessment and understanding of flow throughout the validation process is essential to the elimination of waste. From a validation perspective, optimal flow is created through the process when, for example, users have the ability to automatically create requirements from test scripts for automated traceability thereby eliminating the process of manually tracing each test script to a requirement. Another example is when a user has the ability to navigate through a software application and the test script process is automatically generated. Once generated, it is automatically published to a document portal where it is routed electronically for review and approval. All of this requires AUTOMATION to achieve the principle of FLOW. For process optimization and quality control throughout the validation lifecycle, information should optimally flow throughout the validation process in an efficient manner minimizing process and document bottlenecks with traceability throughout the process.
Principle 4 – PULL – A pull system is a lean manufacturing is used to reduce waste in the production process. Components used in the manufacturing process are only replaced once they have been consumed so companies only make enough products to meet customer demand. There is much waste in the validation process. The PULL strategy for validation may be used to reduce wastes such as duplication of effort, streamlining test case development and execution, electronic signature routing/approval and many others. Check out our blog “The Validation Post” for more information.
Principle 5 – PERFECTION – Validation processes are in constant pursuit of continuous improvement. Automation is KEY. Lean validation engineers and quality professional relentlessly drive for perfection. Step-by-step validation engineers must identify root causes of software issues, anomalies, and quality problems that affect the suitability of a system for production use. As computing systems environments evolve and become more complex and integrated, validation engineers must seek new, innovative ways to verify software quality and compliance in today’s advanced systems. Perfection cannot easily be achieved through manual processes.
Driving Lean Validation Processes
in CRO/CMOs
CRO/CMOs are experiencing an expanding global market. As companies endeavor to do more with less, over the past 20 years, outsourcing has seen a significant surge. Increasingly, companies are outsourcing mission-critical cGxP regulated processes and product manufacturing. Leading companies across the globe rely on Contract Manufacturing Organizations (CMOs) and Clinical Research Organizations (CROs) to improve operational efficiency and productivity.
However, for all its benefits, outsourcing can also present unique challenges to any organization. Global regulatory requirements, GxP compliance, and quality are becoming increasingly more complex as greater risk is assumed by the company. The CRO/CMO business model is not designed to absorb high levels of risk therefore many of them are going LEAN.
Lean Thinking in CRO/CMOs
Lean validation is derived from the lean manufacturing process. Simply put, lean manufacturing is the process of eliminating waste, minimizing risk and driving quality, efficiency and compliance. A common misconception is that lean is suited only for manufacturing. This is not true. Lean principles may be applied to any process that would benefit from the elimination of wasteful activities and process improvement. Lean validation thus is not a tactic or a cost reduction program, but a way of thinking and acting for today’s validation team. The figure below depicts the overall strategy for lean validation.
As highlighted above, lean validation requires automation. ValidationMaster™ powers lean validation and is designed to facilitate automation throughout the process. The system manages the full lifecycle of validation processes and is designed to eliminate traditional manual, paper-based processes.
Mastering Lean Validation
In The Clinical Industry
The increased use of advanced enterprise computer systems by companies within the clinical market sector to generate and maintain source data and source documentation is causing many to think about how this information is managed long term. The U.S. FDA mandates that electronic source data and source documentation meet the same fundamental elements of data quality (e.g., attributable, legible, contemporaneous, original, and accurate) that are expected of paper records and must comply with all applicable statutory and regulatory requirements.
New guidance for data integrity as well as cyber threats are cause for new thinking for clinical organizations across the globe. From a validation perspective, if the systems used by clinical organizations are subject to 21 CFR Part 11, they are subject to validation. Progressive clinical organizations are going lean.
Going Lean in Clinical
Lean validation is derived from the lean manufacturing process. Simply put, lean manufacturing is the process of eliminating waste, minimizing risk and driving quality, efficiency and compliance. A common misconception is that lean is suited only for manufacturing. This is not true. Lean principles may be applied to any process that would benefit from the elimination of wasteful activities and process improvement. Lean validation thus is not a tactic or a cost reduction program, but a way of thinking and acting for today’s validation team. The figure below depicts the overall strategy for lean validation.
There are five principles of lean validation derived from lean manufacturing principles. Lean validation is powered through people, processes and technology. Automation drives lean validation processes.
Principle 1 – VALUE – Lean thinking in manufacturing begins with a detailed understanding of what value the customer assigns to product and services. Lean thinking from an independent validation and verification perspective begins with a detailed understanding of the goals and objectives of the validation process and its adherence to compliance objectives. The principle of VALUE requires the validation team to therefore focus on the elimination of waste to deliver the value the end customer (your organization) in the most cost-effective manner. The computer systems validation process is designed for the purpose of assuring that software applications meet their intended use. The value derived from the validation process is greater software quality, enhanced ability to identify software defects as a result of greater focus and elimination of inefficient and wasteful processes. AUTOMATION IS THE FOUNDATION THAT FACILITATES THE ACHIEVEMENT OF THIS VALUE PRINCIPLE.
Principle 2 – VALUE STREAM – The value stream, from a lean perspective is the comprehensive product life-cycle from the raw materials through customer’s end use, and ultimate disposal the product. To effectively eliminate waste, the ultimate goal of lean validation, there must be an accurate and complete understanding of the value stream. Validation processes must be examined end-to-end to determine what value is added to the objective of establishing software quality and compliance. Any process that does not add value to the validation process should be eliminated. We recommend value stream mapping for the validation process to understand where value is added and where non-value -added processes can be eliminated. Typical “Muda” or wastes commonly revealed from validation process mapping are:
- Wasteful Legacy Processes (“we have always done it this way”)
- Processes That Provide No Value To Software Quality At All
- Manual Process Bottlenecks That Stifle Processes
Principle 3 – FLOW – The lean manufacturing principle of flow is about creating a value chain with no interruption in the production process and a state where each activity is fully in step with every other. A comprehensive assessment and understanding of flow throughout the validation process is essential to the elimination of waste. From a validation perspective, optimal flow is created through the process when, for example, users have the ability to automatically create requirements from test scripts for automated traceability thereby eliminating the process of manually tracing each test script to a requirement. Another example is when a user has the ability to navigate through a software application and the test script process is automatically generated. Once generated, it is automatically published to a document portal where it is routed electronically for review and approval. All of this requires AUTOMATION to achieve the principle of FLOW. For process optimization and quality control throughout the validation lifecycle, information should optimally flow throughout the validation process in an efficient manner minimizing process and document bottlenecks with traceability throughout the process.
Principle 4 – PULL – A pull system is a lean manufacturing is used to reduce waste in the production process. Components used in the manufacturing process are only replaced once they have been consumed so companies only make enough products to meet customer demand. There is much waste in the validation process. The PULL strategy for validation may be used to reduce wastes such as duplication of effort, streamlining test case development and execution, electronic signature routing/approval and many others. Check out our blog “The Validation Post” for more information.
Principle 5 – PERFECTION – Validation processes are in constant pursuit of continuous improvement. Automation is KEY. Lean validation engineers and quality professional relentlessly drive for perfection. Step by step validation engineers must identify root causes of software issues, anomalies, and quality problems that affect the suitability of a system for production use. As computing systems environments evolve and become more complex and integrated, validation engineers must seek new, innovative ways to verify software quality and compliance in today’s advanced systems. Perfection cannot easily be achieved through manual processes.
Going Lean In
The Blood-Tissue Industry
Across the globe in today’s rapid pace environment, regulated companies in the blood and tissue industry are seeking to new technological innovations to address their unique challenges while optimizing processes and efficiency. Compliance is at the very core of every aspect of the business. Risk management ensures that compliance is achieved at each step and thus minimizes potential risks to the patient. The FDA released its publication “Guidance for Industry Blood Establishment Computer System Validation in the User’s Facility”, issued April 2013. The guidance covered software designed to be used in a blood establishment and intended for use in the diagnosis of disease or other conditions in donors, or in the prevention of disease in humans by preventing the release of unsuitable blood and blood components. In summary, the guidance recommends a risk-based approach.
From the standpoint of validation testing, the guidance states “Your records should include documented evidence of all test cases, test input data and test results. Test results should include screen prints. For traceability purposes and to facilitate quality assurance review and follow-up, we recommend that any supporting documentation, such as screen-prints, where appropriate, be identified to link them to the specific test case…”
Capturing the right amount of validation due diligence is key. Given that the Agency would like screen prints as documented evidence as well as traceability, this can be difficult given manual processes.
Today’s forward-thinking blood and tissue organizations are GOING LEAN.
Lean Validation Methodology
Lean validation is derived from the lean manufacturing process. Simply put, lean manufacturing is the process of eliminating waste, minimizing risk and driving quality, efficiency and compliance. A common misconception is that lean is suited only for manufacturing. This is not true. Lean principles may be applied to any process that would benefit from the elimination of wasteful activities and process improvement. Lean validation thus is not a tactic or a cost reduction program, but a way of thinking and acting for today’s validation team. The figure below depicts the overall strategy for lean validation.
As highlighted above, lean validation requires automation. ValidationMaster™ powers lean validation and is designed to facilitate automation throughout the process. The system manages the full lifecycle of validation processes and is designed to eliminate traditional manual, paper-based processes.

Accelerate your IV&V Processes
We bring independence and deep domain experience to each engagement. We specialize in the validation of leading enterprise technologies such as Microsoft Dynamics 365/GP/NAV, Enterprise Content Management, Enterprise Quality Management, LIMS and other quality and compliance related systems.